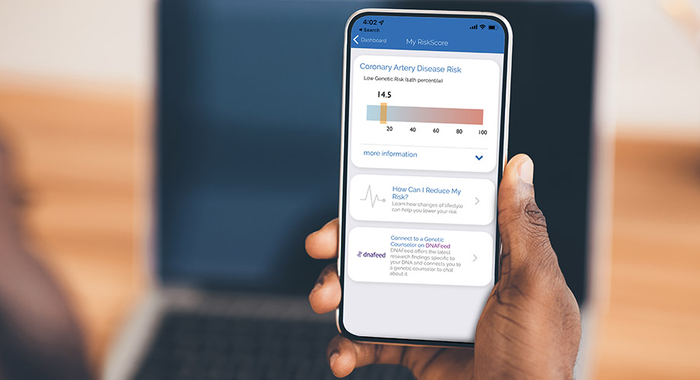
A smartphone app that can calculate a user’s risk of developing coronary artery disease (CAD) encourages at-risk people to take preventative medication, a study has found.
The MyGeneRank app uses genetic information from the 23andMe genetic testing company to calculate the score using DNA data.
In the study, published in npj Digital Medicine, participants with high risk scores were much more likely to start using statins or other cholesterol-lowering therapies, compared to those with low-risk scores.
Study senior author Ali Torkamani, PhD, professor and director of Genomics and Genome Informatics at the Scripps Research Translational Institute, said:
“We saw about twice the rate of statin initiation in the high genetic risk group vs the low genetic risk group, which indicates that strategies like this could make a big contribution to public health—heart disease being the largest cause of death globally.”
Coronary artery disease, also known as coronary heart disease, features the hardening and narrowing of arteries feeding the heart muscle.
In the UK, one in eight men and one in 15 women will die as a result of CAD.
Statins and other non-statin drugs reduce bloodstream levels of cholesterol and other fat-related molecules called lipids.
Increasing use of these drugs has helped lower the annual CAD death rate in the US over the past 20 years.
However, researchers estimate that only a third of 45-65-year-olds who are at risk of CAD take lipid-lowering drugs.
In the study, app-users linked their existing 23andMe data and filled out consent forms and health-related surveys, including questions about the use of lipid-lowering drugs.
The app then calculated and shared with the respondent a risk score based on the latest recognised genetic risk factors for CAD.
The app then followed up approximately a year later with questions on the respondent’s current use of lipid-lowering medications.
Overall, at-risk participants were, at follow-up, about 1.4 times more likely to report use of a statin lipid-lowering drug, and about 4 times more likely to report use of a non-statin lipid-lowering drug.
The high-risk participants also initiated lipid-lowering therapy at an average age of 52, compared to 65 in the low-risk group.
Torkamani said:
“On the whole it looks like a significant effect, especially considering that we were giving respondents only appropriately contextualized information about their gene-based risk scores—we weren’t directly telling them to go out and start taking medications.”
First author Evan Muse, MD, PhD, a cardiologist and lead for cardiovascular genomics at the Scripps Research Translational Institute, added:
“We now have the opportunity to integrate a person’s genetics into their cardiovascular health assessment to help them better understand their individualized risk and empower them to make the necessary modifications – including the addition of statin therapy – to their risk factor optimisation plans.
“Even if someone finds out they have low genetic risk for CAD, knowing their score can help—for example if they also know they have high risk overall, that may suggest other non-genetic factors like lifestyle are contributing to their risk and that they should consider making changes.”








