Every year more than one million people receive a pacemaker.
Until now, leadless versions were only available for 20% of these patients. However, thanks to an international consortium led by Amsterdam UMC and funded by Abbott Medical, an improved version could later this year be available for all patients.
Research from Amsterdam UMC published in New England Journal of Medicine has succeeded in further revolutionising the wireless pacemaker.
The improved dual-chamber version which is smaller than a AAA battery, can now be placed in both the atrium and the ventricle of the heart via a minimally invasive procedure – meaning a larger group of patients can be fitted with a wireless pacemaker.
A decade ago, Amsterdam UMC laid the foundations for the development of the wireless pacemaker.
At the time it was a huge innovation on the traditional pacemaker, that sits above the skin and reaches the heart muscle with a wire.
However, until now, a wireless pacemaker could only be placed in one cavity of the heart: the ventricle. This meant it was only suitable for a small proportion of patients with a slow heart rhythm.
Now, this latest clinical trial – known as the AVEIR dual-chamber (DR) i2i Investigational Device Exemption (IDE) study – has shown that a device can also be implanted in the atrium of the heart.
The AVEIR-DR has been designed by Abbott with a first-of-its-kind i2i (implant-to-implant) technology with the goal of providing beat-to-beat communication and synchrony between two leadless pacemakers. Unlike other leadless pacemakers, this system allows the two devices to communicate with each other, sensing for delayed or missed heartbeat and then pacing the appropriate chamber of the heart.
In addition, Abbott’s AVEIR leadless devices utilise specially designed attachment mechanisms that allow for retrieval of the pacemakers when changes in therapy are needed.
In the AVEIR DR i2i IDE study, physicians successfully implanted leadless pacemakers in the right atrium along with one inserted in the right ventricle for the first time.
Lead researcher Reinoud Knops, professor of electrophysiology at Amsterdam UMC, said: “Most patients need a pacemaker that works in both the atrium and the ventricle for optimal contraction of the heart. Before now, that wasn’t possible as it is very complicated to place two mini pacemakers that can communicate with each other wirelessly.
“After thorough research and testing, we managed to make it possible. This means that those who need a pacemaker will soon be able to count on a new treatment.”
The new pacemakers communicate with each other via electrical pulses and were implanted for the first time in 300 people who were then followed for a minimum of three months. The results of this study showed that the treatment is safe, and that the system works well.
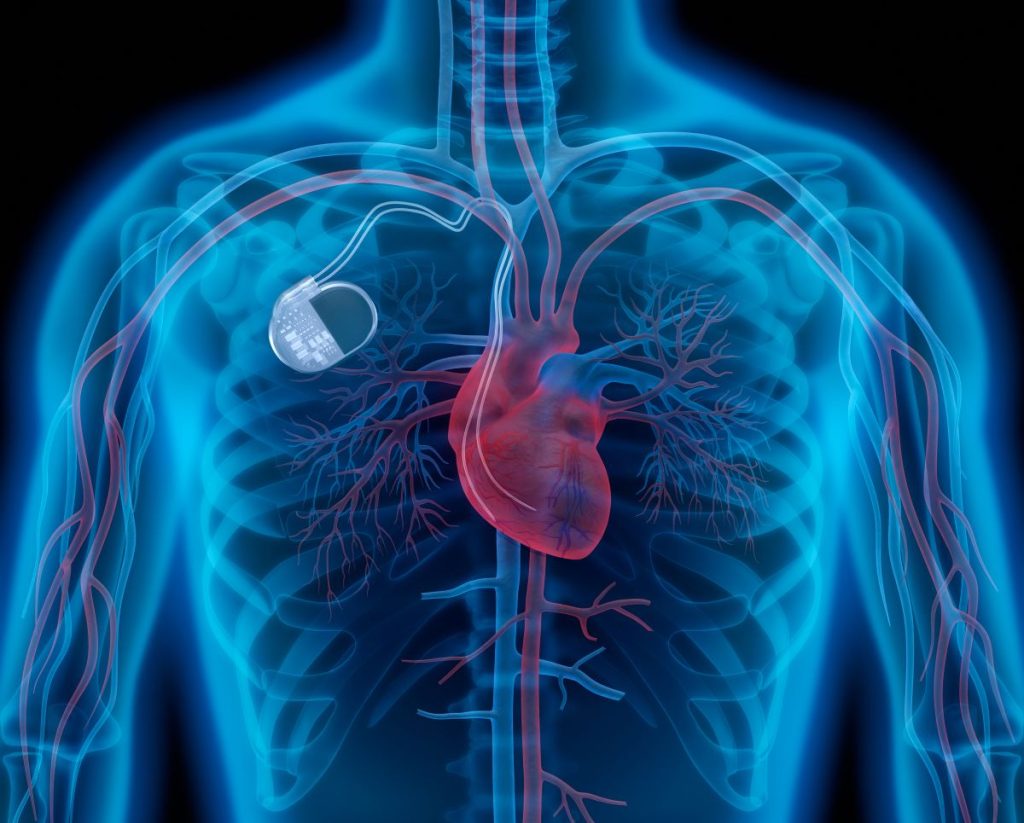
Pacemakers have been a basic treatment for patients with a slow heart rhythm for many years.
Traditional pacemakers consist of a subcutaneous box under the collarbone with a wire connected to the heart by a vein.
But these cables are fragile and can break, become detached from the heart, or become infected. This can lead to patients having to go back to the hospital for another operation.
For this reason, Amsterdam UMC developed its mini pacemaker without a box or wiring ten years ago, which is the size of a vitamin.
Abbott Medical has announced data from the clinical trial at the same time as Amsterdam UMC and presented its findings at the Heart Rhythm Society’s 44th annual meeting held in New Orleans.
The Chicago-headquartered multinational has also now provided data from the trial to the US Food and Drug Administration (FDA) as part of its submission for approval of the AVEIR device. It is hoped it will get the nod in the US in the third quarter of 2023 and in Europe in 2024.