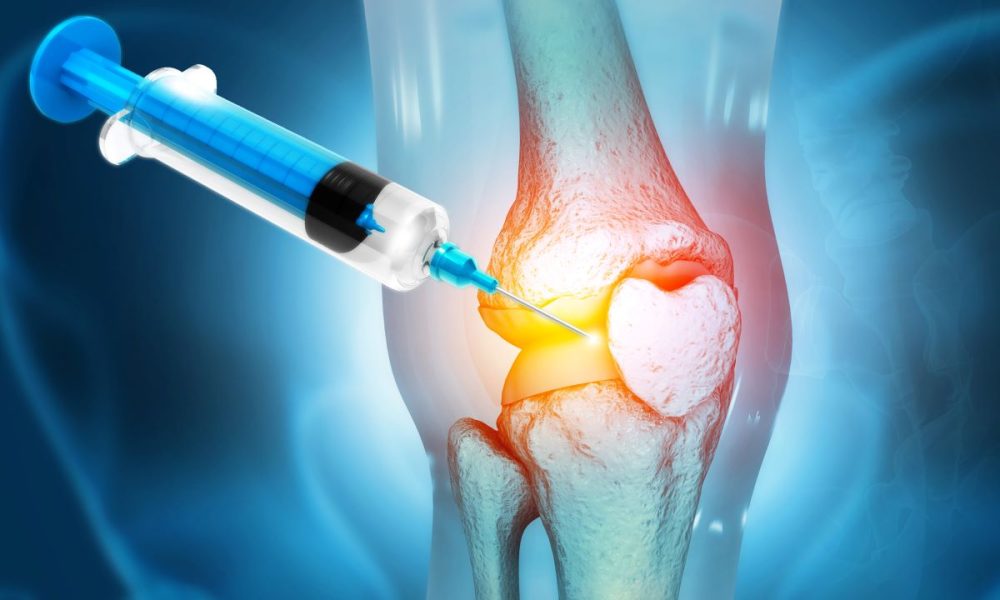
A new, injectable nanotechnology-based treatment for arthritis – hydrocelin (ALG-001) – has shown positive safety results in three preclinical studies.
The studies found no signs of pyrogenicity, irritation or delayed sensitisation in the test groups, strengthening hydrocelin’s safety profile after two earlier preclinical studies announced in December.
Allegro, which has developed the microparticle hydrogel, has said it intends to launch a multi-centre pivotal trial in osteoarthritis patients in Belgium later this year.
“These preclinical results are an important step in fulfilling Allegro’s mission of giving back mobility to the millions of people suffering from osteoarthritis around the world,” said Lucas Decuypere, CEO at Allegro.
“Allegro’s hydrocelin injection, which we expect will need to be administered once a year, is designed to provide immediate pain relief for patients and deliver disease-modifying therapeutic benefit. With an ageing population, the need for a lasting solution for osteoarthritis will continue to grow in importance.”
Hydrocelin contains cross-linked microparticles acting as tiny shock absorbers in the synovial fluid of the joints.
Restoring the shock-absorbing capacity of the synovial fluid reduces the force on sensitive nerve endings, providing immediate pain relief, and potentially protecting cartilage.
In the preclinical studies, hydrocelin was shown to be safe to use in three important aspects: the potential to cause irritation following intracutaneous injection, pyrogenicity and delayed sensitisation.